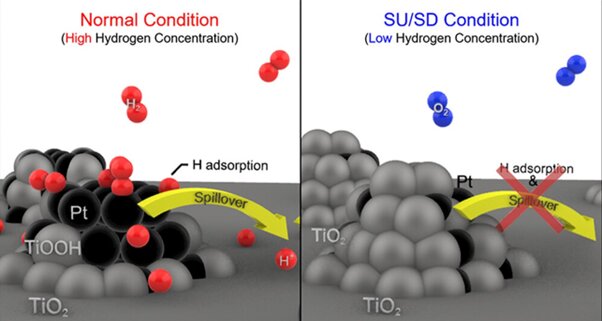
<बाएं> The given figure shows hydrogen spillover, in which TiO2 Under high hydrogen concentration conditions TiOOH undergoes conformational changes, which promote hydrogen mobility on the surface and consequently generate conductivity.2 The covering of Pt by TiO leads to2, Credits: Postech
Professor Yong-Tae Kim (Department of Materials Science and Engineering and Graduate Institute of Iron and Energy Materials Technology) and Ph.D. A research team led by candidate Sang-Hoon Yoo (Department of Materials Science and Engineering) at Postech has developed a selective catalyst that inhibits corrosion in fuel cells used for hydrogen-powered automobiles.
By optimizing the hydrogen oxidation reaction to match the concentration of hydrogen in the fuel cell, the team was able to prevent corrosion of the fuel cells. Posted in research ACS Energy Sheet,
Fuel cells are susceptible to a number of factors that degrade their durability. One of them is degradation, especially in the cathode catalyst, which is regularly exposed to start-up and shut-down events in automobiles. In particular, fuel cells designed for automotive use inherently experience recurring cycles of start-up and shut-down.
During normal vehicle operation, hydrogen is continuously supplied with high concentrations to the fuel cells, but when the car is turned off or started the hydrogen concentration temporarily decreases. As a result, when outside air mixes with the hydrogen within the fuel cells, an unintended oxygen reduction reaction is triggered at the anode, causing a sudden potential jump and carbon corrosion at the cathode.
The research team developed a catalyst (Pt/TiO2), containing platinum (Pt) titanium dioxide (TiO2), which efficiently prevents corrosion in fuel cells employed in hydrogen-powered automobiles. The performance of this electrocatalyst comes from the strong interaction between titanium dioxide and platinum, and from the ability of hydrogen spillover to modify the material’s surface conductivity in response to the hydrogen concentration around it.
When a vehicle stops or starts suddenly, the concentration of hydrogen within the fuel decreases accordingly. As a result of this decrease in hydrogen concentration, titanium dioxide expands on the platinum, resulting in the platinum being buried below the surface of the catalyst.
This burial of platinum due to expansion of titanium dioxide eventually turns the catalyst into an insulator due to the low conductivity of titanium dioxide. This insulating effect hinders the catalyst’s ability to conduct electricity, thus preventing unwanted depletion of oxygen that can cause a sudden potential jump at the cathode.
In contrast, during a standard vehicle operation, the concentration of hydrogen inside the car remains high. Under such high hydrogen concentration conditions, the highly conductive platinum is exposed on the surface of the catalyst, and the reduction of titanium dioxide, which promotes hydrogen mobility on the surface of the catalyst. This phenomenon, called hydrogen spillover, increases current flow and accelerates the hydrogen oxidation reaction.
The research team also conducted a simulation test to compare the newly developed catalyst and conventional catalysts. Test results demonstrated that fuel cells using Pt/TiO2 The catalyst displayed three times greater durability relative to conventional fuel cells. This indicates that the team has successfully enhanced the durability of fuel cells through the use of a hydrogen oxidation reaction based on a selective oxygen reduction reaction and a hydrogen oxidation reaction.
If this research can contribute to overcoming the current sustainability challenges facing fuel cells for hydrogen-powered vehicles, it could potentially enhance the position of Korean hydrogen-fueled automobiles in the next-generation mobility industry. .
more information:
Sang-hoon Yoo et al, Enhanced durability of automotive fuel cells through selective implementation by hydrogen spillover on the electrocatalyst surface, ACS Energy Sheet (2023). DOI: 10.1021/acsenergylett.2c02656
Citation: Extending fuel cell lifetime through catalyst selection (2023, 12 May) retrieved 14 May 2023
This document is subject to copyright. No part may be reproduced without written permission, except in any fair dealing for the purpose of private study or research. The content is provided for information purposes only.

/cdn.vox-cdn.com/uploads/chorus_asset/file/24016885/STK093_Google_04.jpg)


/cdn.vox-cdn.com/uploads/chorus_asset/file/24808816/Starfield__The_Settled_Systems___Supra_Et_Ultra_____Starfield__The_Settled_Systems___Supra_Et_Ultra_2023_7_25_94252.263_1440p_streamshot.png)